News
Medicine Grand Rounds on May 14, 2025
Medicine Grand Rounds Hosted via Zoom
May 14, 2025 12:00 pm – 1:00 pm
Jerome Federspiel, MD, PhD
Assistant Professor of Medicine-Hematology
Duke University School of Medicine
Photo courtesy of Duke Health
Pregnancy and Blood Disorders: Better Together
Learning Objectives:
- Understand the potential impacts of pregnancy on blood disorders, and of blood disorders on pregnancy.
- Have competency counseling patients with blood disorders who are completing pregnancy.
- Approach blood disorders in pregnancy from a structured, multidisciplinary fashion.
- Improve the maternal and fetal pregnancy outcomes of pregnancies complicated by blood disorders.
To join via Zoom please contact emcdowe3@uthsc.edu
Disclosures:
Matria Health Solutions, Noma.AI
1 CME/CEU credit provided


The University of Tennessee College of Medicine is committed to providing CME that is balanced, objective, and evidence based. In accordance with the Accreditation Council for Continuing Medical Education (ACCME) Essentials and Standards, all parties involved in content development are required to disclose any real or apparent conflicts of interest relating to the topics of this educational activity. The University of Tennessee College of Medicine has established mechanisms to resolve conflicts of interest should they arise. Any potential conflict of interest or off label use will be disclosed to the audience prior to the activity.
AMA Credit Designation: The University of Tennessee College of Medicine (UTCOM) designates this live activity for a maxium of 1 AMA PRA Category 1 CreditsTM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Continuing Education for Non-Physicians: The UTCOM will issue Certificates of Participation to non-physicians for participating in this activity and designates it for CEUs using the national standard that 1 hour of educational instruction is awarded 1 CEU.
Accreditation: The UTCOM is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Congratulations to Dr. Ogu on her MSK SCD Symposium!
Congratulations to Dr. Ogu on her MSK SCD Symposium, "Advances in the Treatment of Sickle Cell Disease: Moving Toward a Cure", at which she will be speaking as an invited faculty.
https://mskcc.cloud-cme.com/course/courseoverview?P=5&EID=49354
Congratulations to Dr. Ogu on her new publication!
Congratulations to Dr. Ogu on her new publication, "Racial disparities in cardiovascular manifestations among patients with sickle cell trait: analysis of national inpatient sample data (2016–2020)".
https://link.springer.com/article/10.1007/s00277-025-06306-0
http://dx.doi.org/10.4324/9781003463931-16
Congratulations to the Sickle Cell group on their publication, "Persistent albuminuria and chronic kidney disease in adults with sickle cell anaemia: Results from a multicenter natural history study."
Zhou LY, Derebail VK, Desai PC, Elsherif L, Patillo KL, McCune P, Wichlan D, Landes K, Ogu UO, Nelson M, Loehr LR, Cronin RM, Tang Y, Cai J, Ataga KI. Persistent albuminuria and chronic kidney disease in adults with sickle cell anaemia: Results from a multicenter natural history study. Br J Haematol. 2024 Jul 8. doi: 10.1111/bjh.19636. Online ahead of print. PMID: 38978309
Congratulations to the Sickle Cell group on their publication, "Association of biomarkers of endothelial function, coagulation activation and kidney
injury with persistent albuminuria in sickle cell anaemia."
Elsherif L, Tang Y, Patillo KL, Wichlan D, Ogu UO, Landes K, McCune P, Scott LC, Gulledge
W, Woodland WH, Nelson M, Loehr LR, Cronin RM, Desai PC, Zhou LY, Pollock DM, Zou
F, Cai J, Derebail VK, Ataga KI. Association of biomarkers of endothelial function,
coagulation activation and kidney injury with persistent albuminuria in sickle cell
anaemia. Br J Haematol. 2024 Sep 3. doi: 10.1111/bjh.19743. Online ahead of print.
PMID: 39228027
Using Disease Modifying Therapies in Sickle Cell Disease" at an educational session at the 65th annual meeting of the American Society of Hematology in San Diego, CA on December 11, 2023.


Congratulations to Dr. Keeneth Ataga, Dr. Jane Hankins, and Ming Chen, MS on their recent publication, “Age-related differences in risks and outcomes of 30-day readmission in adults with sickle cell disease.”
Congratulations to Dr. Kenneth Ataga on his recent publication, “Evaluation of treatment patterns, healthcare resource utilization and cost of illness for sickle cell disease in Ghana: a private medical insurance claims database study.”
The Memphis Business Journal hosted their 2023 Health Care Heroes Awards on Thursday, August 17, 2023. This event recognized and honored organizations, institutions, and individuals throughout the city of Memphis who have made great contributions in the healthcare field. Dr. Kenneth Ataga was a finalist for the Health Care Innovations award.
Congratulations Dr. Ataga on your nomination!
Congratulations to Dr. Keesha Roach, an early career Nurse Scientist at the University of Tennessee Health Science Center. Her daughter recently got married and completed her PhD in Animal Science at Iowa State last month. Way to go!
Ramyla Dahmer, a high school senior living with sickle cell, was presented with the 2023 Dr. Patricia Adams-Graves scholarship.



Winer JC, Yee ME, Ataga KI, Lebensburger JD, Zahr RS. Br J Haematol. 2022 Oct 3. doi: 10.1111/bjh.18494. Online ahead of print. PMID: 36191963 No abstract available.
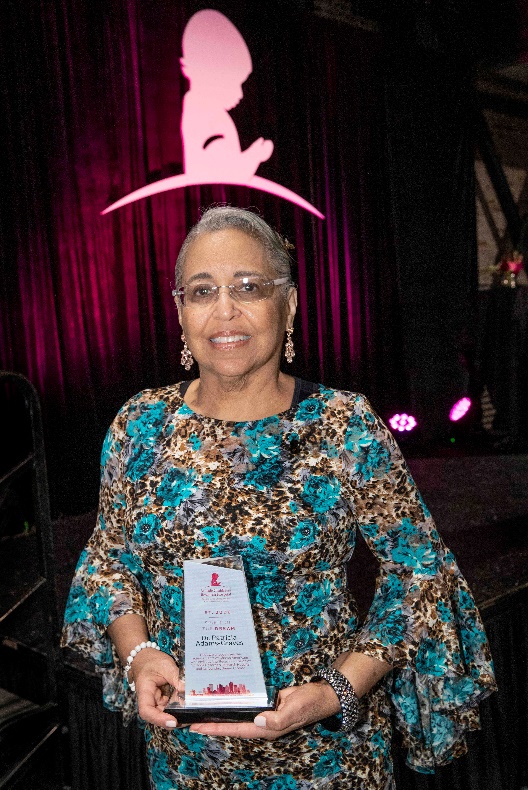
Congratulations to Dr. Patricia Adams-Graves, a professor in the Hematology/Oncology Division at the University of Tennessee Health Science Center. She was presented with the Spirit of the Dream award by St. Jude on September 29, 2022 for the impact she has made serving and caring for those living with Sickle Cell Disease in the greater Midsouth region.

Dr. Ataga is the Scientific Chair for the Foundation for Sickle Cell Disease Research’s September Symposium on Machine Learning, which will occur virtually on September 22, 2022 from 10 am – 4 pm EST. Register for the Symposium.


Hematology Speaker Series: New Approaches for Transfusion Support of Sickle Cell Disease
Save the Date!
October 19, 2022
12:00 pm – 1:00 pm
Join us via Zoom:
Meeting ID: 985 3966 3746
Passcode: 965370
Stella Chou, MD
Chief, Division of Transfusion Medicine, The Children's Hospital of Philadelphia Associate
Professor of Pediatrics, the Children's Hospital of Philadelphia University of Pennsylvania School
of Medicine
1 CME/CEU credit provided
AMA Credit Designation: The University of Tennessee College of Medicine (UTCOM) designates this live activity for a maxium of 1 AMA PRA Category 1 CreditsTM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Continuing Education for Non-Physicians: The UTCOM will issue Certificates of Participation to non-physicians for participating in this activity and designates it for CEUs using the national standard that 1 hour of educational instruction is awarded 1 CEU.
Accreditation: The UTCOM is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Sponsored by the Methodist Foundation

Congratulations to Dr. Jane Hankins on her recent publication!
Longitudinal study of glomerular hyperfiltration in adults with sickle cell anemia:
a multicenter pooled analysis. Ataga KI, Zhou Q, Saraf SL, Hankins JS, Ciccone EJ, Loehr LR, Ashley-Koch AE, Garrett
ME, Cai J, Telen MJ, Derebail VK; Blood Adv. 2022 August 9.

Congratulations to Dr. Patricia Adams-Graves for being selected to receive the 2022 St. Jude Spirit of the Dream Award presented by ALSAC. This award recognizes the achievements of an African-American who embodies the lifesaving mission of St. Jude Children’s research Hospital and its founder, Danny Thomas.
We also congratulate her for an honor received earlier this year with a scholarship in her name. The Dr. Patricia Adams-Graves Sickle Cell Scholarship was created in collaboration with the Sickle Cell Foundation of Tennessee (SCFTN) and The Memphis Alumnae Chapter of Delta Sigma Theta Sorority, Inc. This scholarship provides financial support toward the pursuit of higher education for high school seniors who have achieved academic excellence while living with sickle cell disease.
Dr. Adams-Graves is deserving of these honors due to her years of trail-blazing advocacy for those living with sickle cell disease. Her work to implement the first sickle cell disease-specific hospital unit in the nation and a computer education curriculum for nurses who deliver care for sickle cell utilized across the nation have positively impacted the lives of countless individuals with this disease, their families, and their caregivers.
Dr. Marquita Nelson was listed in Memphis Magazine's 2022 “Top Doctors” list.
Dr. Ataga's article, "Longitudinal Study of Glomerular Hyperfiltration in Adults with Sickle Cell Anemia: A Multicenter Pooled Analysis" has been published online by ScienceDirect.
Kenneth Ataga, MD, Director of the Center for Sickle Cell Disease at the University of Tennessee Health Science Center (UTHSC), was recently awarded $3.2 million from the National Heart, Lung and Blood Institute for a project that enlists artificial intelligence in the fight against sickle cell disease. Santosh Saraf, MD, associate professor in the Department of Medicine at the University of Illinois at Chicago, is a co-PI on the grant. (Read More)
A recent paper from our group on machine learning and prediction of rapid kidney function decline in sickle cell anemia. (Read the latest publication)
One of the missions of the Department of Medicine is to foster and develop research. In efforts to support research and the development of new extramural funding, the DOM has offered 2 Development Grants for this upcoming academic year for faculty in the Department of Medicine. It is anticipated that this award will culminate in a new grant application by the end of the academic year. We received many excellent applications for these awards that were peer reviewed by a committee. I am pleased to announce that the following faculty were selected for funding this year:
Laila Elsherif, PhD
Assistant Professor of Medicine
Division of Hematology & Oncology
“Effect of PAI-1 Increase on Neutrophil Apoptosis Delay in Sickle Cell Disease”
Zhousheng Xiao, PhD
Associate Professor of Medicine
Division of Nephrology
“Novel Role of PC1/PC2 Complex in Adipocyte Metabolism”
Please help me in congratulating our Department of Medicine grant awardees!
This article was originally published on UTHSC News. You can view the original news article here.
Written by Lee Ferguson | August 4, 2020
A new research project that could impact and expand the discovery of new treatment options for sickle cell patients has received significant federal funding. The National Heart, Lung, and Blood Institute awarded nearly $1 million to Athena Starlard-Davenport, PhD, assistant professor of Genetics, Genomics, and Informatics in the UTHSC College of Medicine, for her project titled, “MicroRNA-based epigenetic approach to induce fetal hemoglobin.”
The grant, the first national award for Dr. Starlard-Davenport, is also a significant milestone for UTHSC’s Office of Research, as it puts the total external dollars generated from CORNET-funded work over the $20 million mark. The CORNET Awards are a seed funding program for interdisciplinary collaborations on new research.
“For the past 100 years, only four drugs have been approved by the FDA for the treatment of sickle cell disease,” Dr. Starlard-Davenport said. “Those patients need more treatment options depending on their symptoms and complications.”
Sickle cell disease is very common. One in 13 African Americans have the sickle cell trait and over 100,000 people in the U.S. have sickle cell disease. Individuals with sickle cell suffer from severe pain crises that increase their risk of organ failure and premature death. The sickle cell mutation causes production of abnormal sickle hemoglobin during the first year of life, when the body makes its switch from producing fetal hemoglobin (gamma globin) to adult beta globin. Fetal hemoglobin is normal in people with sickle cell disease; it is only the adult form of sickle hemoglobin that is abnormal. Scientists have found that reversing this normal switch, i.e. turning the gamma globin gene back on, counteracts symptoms, improves health outcomes and prolongs the lifespan of sickle cell patients.
To date, hydroxyurea is the only FDA-approved drug with proven efficacy to do that. Though another class of epigenetic modulators − DNA methyltransferase (DNMT) inhibitors – has shown promise inducing fetal hemoglobin in sickle cell patients, they produce off-target side effects. Dr. Starlard-Davenport’s research is focused on a specific small molecule in this class, miRNA29b, for further study.
Dr. Starlard-Davenport has been investigating miRNA29b and fetal hemoglobin induction for four years. She got her start in 2016 with help from a new seed funding initiative launched that year by the UTHSC Office of Research, the CORNET Awards. She and her collaborator, Patricia Adams-Graves, PhD, professor of Medicine and hematologist at the Diggs-Kraus Sickle Cell Clinic at Regional One Health, won a CORNET in the Clinical Awards division for their project, “Investigation of miR29 as a novel fetal hemoglobin inducer and treatment for patients with sickle cell disease.”
“The CORNET award provided funding support to collect blood samples from sickle cell patients and to test whether our miRNA therapeutic could increase levels of fetal hemoglobin in vitro in cell culture and in preclinical sickle cell models. We did this work in collaboration with Betty Pace at Augusta University,” Dr. Starlard-Davenport said. Betty Pace, MD, is the Tedesco Distinguished Chair of Pediatric Hematology/Oncology at Augusta University. “The funding was used to purchase reagents for running the necessary biochemical assays, including our miRNA mimic, cell culture reagents.”
The CORNET program was begun by Steven Goodman, PhD, vice chancellor for Research, to encourage interdisciplinary collaboration on new research that will lead to larger, national grants. In the nearly four years since their inception, the awards have supported 52 new collaborative research teams and their groundbreaking initiatives. With this latest national award, external dollars generated from CORNET work has reached $20,604,972, an 11.6-fold return on a $1.78 million investment.
“This NIH award for studies to increase fetal hemoglobin expression thereby reducing sickle cell severity is important for many overlapping reasons,” Dr. Goodman said. “It is the first NIH award for Dr. Starlard-Davenport. We were able to assist Athena and her colleagues to obtain the preliminary data that led to the award through the CORNET award program. This extramural award put the CORNET program over the $20 million threshold in return on investment. Dr. Starlard-Davenport has been a mentee in the wonderful PRIDE program, which focuses on jump-starting the career of junior underrepresented minority faculty. The director of this PRIDE Program is Dr. Betty Pace who mentored Dr. Starlard-Davenport and is an investigator on the NIH Award. It is amazing how many stars aligned in this one NIH award to a very deserving new investigator. But most important is the impact that this study can have upon the quality of life of people around the globe who suffer from this genetic disease.”
“In the initial CORNET application, my team provided preliminary results to show that miRNA29b could increase levels of fetal hemoglobin in a compatible human cell line, but we needed further preliminary data to determine what effect miRNA29b had on fetal hemoglobin induction in red blood cells from individuals with and without sickle cell disease,” Dr. Starlard-Davenport said. The NIH funding she just received, $928,317 to be distributed over three years, will allow her team to collect blood samples from sickle cell patients and test whether this molecule increases the expression of fetal hemoglobin that blocks sickle hemoglobin polymerization and improves clinical symptoms. The long-term goal is to develop miRNA29b as a new drug for treating patients with sickle cell.
Dr. Starlard-Davenport is the principal investigator on a team of clinicians and scientists that includes, in addition to Dr. Adams-Graves and Dr. Pace, Ken Ataga, MD, director of the UTHSC Center for Sickle Cell Disease; Daniel Johnson, PhD, director of the UTHSC Molecular Bioinformatics Center; and Biaouri Li, MD, professor in the department of Pediatric Hematology/Oncology at Augusta University.
This article was originally published on UTHSC News. You can view the original news article here.
View a video recording of the 2019 Norfleet Forum at the following links. Part 1 | Part 2





Clinicians and researchers from around the country gathered in Memphis Friday for the 2019 Frank M. Norfleet Forum for the Advancement of Health to discuss sickle cell disease research and treatment. They looked back at how the disease has developed over the last century and what the future holds for those born with it and those who are trying to cure it.
Themed “More than 100 years of Sickle Cell Disease: Development of Novel Therapies,” the daylong conference at the National Civil Rights Museum was organized and led by Kenneth Ataga, MD, director of the Center for Sickle Cell Disease at the University of Tennessee Health Science Center and the Memphis Consortium for Sickle Cell Disease and Non-Malignant Hematology Research. Dr. Ataga joined UTHSC in 2018 and is recognized nationally and internationally as an expert clinician and researcher in the field of sickle cell disease.
Progress has been made in diagnosing and treating sickle cell disease, the panelists said.
Improvements in drug therapies, screening for the disease at birth, better supportive care, developing gene therapy, and ongoing research have improved outcomes for those with the disease.
“A lot of work still has to be done,” Dr. Ataga told the group.
He said it is necessary to improve access to comprehensive care by specialists, increase advocacy for those with the disease, improve the infrastructure for clinical trials, and promote drug discovery and testing of novel compounds to treat the disease.
Health disparities, including poverty and lack of access, prevent many patients around the world from getting the care that could improve their quality of life.
Panelists included researchers from Boston Children’s Hospital, Albert Einstein College of Medicine, Duke Comprehensive Sickle Cell Center, St. Jude Children’s Research Hospital, Cincinnati Children’s Hospital, and Vanderbilt University.
The first Norfleet Forum was held in 1980 at UTHSC. The annual conference was created to focus on the improvement of health in the community, the state, and around the world through discussion of health policies and developments. It was funded by a gift to the university in honor of Memphis businessman Frank M. Norfleet through the Community Foundation of Greater Memphis. Other recent topics have included kidney disease in the African American community, childhood obesity, and alcohol use disorders.
Jon McCullers, MD, chair of pediatrics at UTHSC and pediatrician in chief at Le Bonheur Children’s Hospital, said the Norfleet Forum allows the university to improve scientific and community knowledge on relevant health care issues and to foster collaboration among researchers that could result in moving science forward.
“Norfleet allows us to bring in 10 or 15 outside speakers, which is a tremendous thing that you don’t see at a local or regional conference,” he said. “It’s unique and it’s very much appreciated. “
FOX13 Memphis – Story by Kody Leibowitz, July 9, 2019
The University of Tennessee Health Science Center College of Medicine's Executive Dean Scott Strome announced the award of internal funds to four projects in the College of Medicine at Memphis.
For the Collaborative Intramural Research in the College of Medicine (CIRCA) support program the following were selected for funding:
The Association of Podocyturia with Progressive Glomerular Injury in Sickle Cell Disease
o Kenneth I. Ataga, MD, Plough Foundation Endowed Chair & Professor of Medicine and Pediatrics
o Adebowale Adebiyi, PhD, Associate Professor, Department of Physiology
The goal of the CIRCA program is to stimulate innovative, interdisciplinary, team research leading to submission of an R01-scale grant application. Specifically, CIRCA is designed to fund innovative research collaborations between a clinician (patient care responsibilities and/or a clinical researcher) and basic researcher in different departments. Matthew Ennis, PhD, Chair of the CIRCA proposal review committee noted “There were many meritorious applications and the decision to fund 2 proposals was an excellent choice based on thorough review by a committee with broad research expertise."
This article was originally published on UTHSC News. You can view the original news article here.
Written by Sarah Ashley Bloch | June 26, 2019
Newly published results from a multinational study co-authored by Kenneth Ataga, MD, director of the Center for Sickle Cell Disease at the University of Tennessee Health Science Center (UTHSC) and the Memphis Consortium for Sickle Cell Disease and Non-Malignant Hematology Research, indicated voxelotor, a novel experimental drug for sickle cell disease, reduced anemia and boosted the health of red blood cells in patients. Dr. Ataga is also the Methodist Endowed Chair in Sickle Cell Anemia at Methodist Le Bonheur Healthcare.
Sickle cell disease is an inherited blood disorder that affects red blood cells. It is caused by a genetic mutation in hemoglobin, resulting in the formation of abnormal hemoglobin known as sickle hemoglobin (HbS). Deoxygenation and the sickling of red blood cells can lead to chronic anemia and hemolysis, and ultimately result in multiorgan damage, stroke, and premature death. More than 100,000 Americans are affected by sickle cell disease, and Memphis has one of the largest populations for adult sickle cell disease in the country. There are only two U.S. Food and Drug Administration-approved treatment options currently available.
“With the limited drug therapies available for patients with sickle cell disease, the findings of this trial are quite exciting,” said Dr. Ataga, who also serves as the Plough Foundation Endowed Chair in Sickle Cell Disease. “With the complex nature of sickle cell disease, the development of drugs that have different mechanisms of action offers an opportunity for combination drug therapy and for individualized therapy based on the presence of specific disease-related complications.”
The Biopharmaceutical company Global Blood Therapeutics, Inc. aims to bring voxelotor to the market as a third treatment option.
Designed to evaluate the efficacy and safety of voxelotor, the phase 3 Hemoglobin Oxygen Affinity Modulation to Inhibit HbS PolymErization (HOPE) Study enrolled 274 patients, ages 12 to 65, with sickle cell disease, from 60 institutions across 12 countries. The patients were divided into three trial groups that received either a 900-mg or 1,500-mg daily dose of voxelotor or a placebo. Clinical assessments were performed at various time points throughout the trial.
The study found that 51 percent of the participants who were administered the higher dose of voxelotor experienced a significant increase in hemoglobin levels after six months of treatment compared to seven percent in the placebo group. In addition, voxelotor treatment reduced the incidence of worsening anemia and the amount of hemolysis. Voxelotor was also deemed safe and well-tolerated by participants.
“While voxelotor improved anemia in the study patients, more studies are required to show whether there are also improvements in other sickle cell disease-related complications,” Dr. Ataga said.
Longer-term follow-up studies are being planned to assess voxelotor’s effects on vaso-occlusive crisis (or sickle cell crisis), morbidity, and mortality in sickle cell patients.
The HOPE Study results were recently published in The New England Journal of Medicine and presented at the European Hematology Association 24th Congress in Amsterdam.
Hematology Speaker Series
| Name | Institution | Title | Date |
| Jerome Federspiel, MD, PhD | Duke University Medical Center | Pregnancy and Blood Disorders: Better Together | 5/14/25 |
| Kenneth Ataga | University of Tennessee Health Science Center | Advancing Academic Hematology at UTHSC | 9/11/24 |
| Marquita Nelson, MD | University of Tennessee Health Science Center | Systems-Based Hematology | 5/8/24 |
| Ulrike Reiss, MD | St. Jude Children's Research Hospital | Hemophilia - Current Therapy Options And Outcomes | 11/29/23 |
| Stella Chou, MD | Children's Hospital of Philadelphia | New Approaches for Transfusion Support of Sickle Cell Disease | 10/19/22 |
| Stephan Moll, MD | University of North Carolina at Chapel Hill | 2022 Clinical Update on DVT, PE, Anticoagulation and Thrombophilia | 1/19/22 |
| Akshay Sharma, MD | St. Jude Children's Research Hospital | Hematopoietic Cell Transplantation and Gene Therapy for Sickle Cell Disease | 10/15/21 |

