Marko Radic, PhD

Associate Professor
858 Madison Ave.
Room 201 Molecular Science Building
Memphis, TN 38163
mradic@uthsc.edu
Phone: 901.448.8219
Fax: 901.448.7360
Education
- PostDoc, Institute for Cancer Research, Fox Chase Cancer Center, Philadelphia, PA, Structure-Function Analysis of Anti-DNA Autoantibodies
- PhD, University of California at Irvine, Irvine, CA, Biological Sciences
- BS, University of California at Davis, Davis, CA, Genetics
Research Interests
Research Description
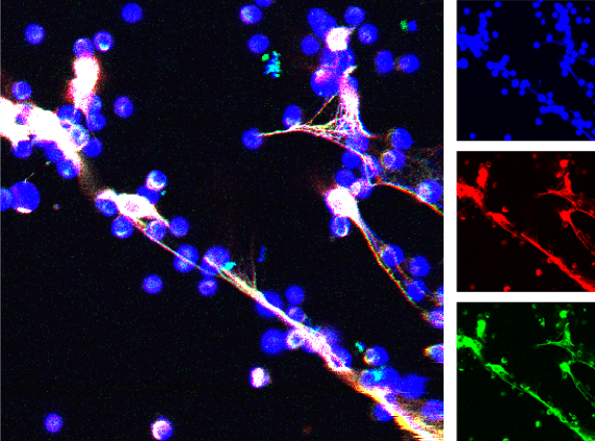
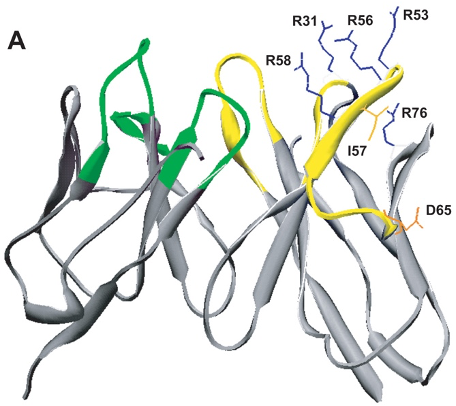
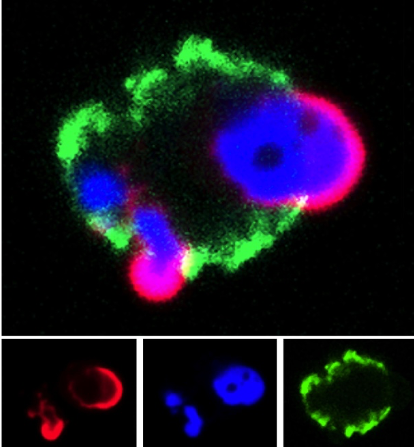
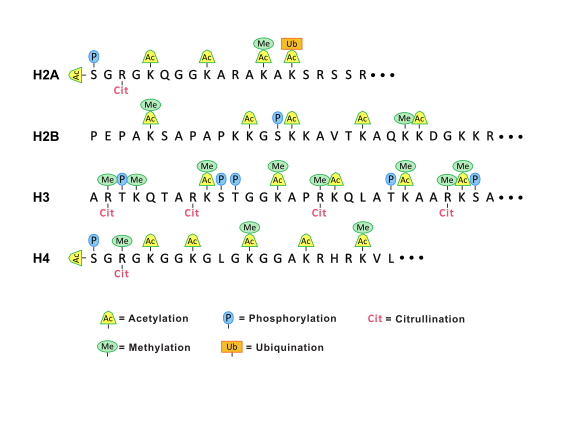
Research in the Radic lab seeks to uncover how the structure of an autoantibody can teach us about the initial steps in autoimmunity. This is because immunity to self follows many of the principles that apply to foreign antigens, yet only rare events successfully overcome self-tolerance. This insight led us to examine how autoantigens are externalized during cell death and engage with receptors on B cells. In continued studies, we found that activation of peptidylarginine deiminase (PAD4) by inflammatory stimuli generates deiminated histones, which represent preferential autoantibody targets. This finding suggested a direct molecular link between inflammation and the induction of autoantibodies, which is a crucial step toward self-sustaining autoimmunity. The implications of these studies were that one critical nexus in the development of lupus lies in the sustained activation of autoreactive B cells. To decisively attack these B cells, we decided to test immunotherapy with anti-CD19 CAR-modified CD8+ T cells in two murine models of lupus. The results of the initial studies, funded by the Lupus Research Alliance, provided very encouraging, recently published data. In the NZB x NZW F1 and MRL-lpr mouse models, we observed that administration of mouse CD19-directed syngeneic CD8+ T cells suppressed the endogenous CD19+ B cell development and strongly reduced autoantibody production. More importantly, this effect persisted over a period of several months, while autoimmune pathology was largely inhibited and even, remarkably, reversed. Our ongoing research seeks to extend these studies and further develop this unique experimental system. We hope, these experiments will set the scene for future application of CAR T cells in lupus.
Research Interest
Molecular Mechanisms of Autoimmune Diseases and Discovery of Cures for Lupus
Research Keywords
Autoantibodies,
Autoimmunity,
B Cell Development,
Chimeric antigen receptors (CARs),
Cytotoxic T cells,
Histones,
Neutrophils,
Post-translational modifications
